PADMA products are based on the knowledge and inspiration of complex multi-substance formulas from Tibet. The interplay of perfectly harmonised ingredients of plant and mineral origin determines their effectiveness. Only high-quality and carefully tested materials are used in PADMA capsules.
The effect of the original PADAM formulas is based on the quality of the raw materials and the gentle technology that preserves the power of the plants and minerals. PADMA deliberately avoids the use of unnecessary synthetic additives. Nature is perfect, why improve it?
Plants
The scarcity of resources requires a special approach. In order to keep the ecological footprint as small as possible, PADMA strives to source as many raw materials as possible from Switzerland and neighbouring countries. Where this is not possible in Europe, sourcing is carried out from specialised cultivation on other continents, where PADMA strives for long-term partnerships with trusted producers.

Not every plant species can be successfully cultivated. For some plants, controlled wild collection is therefore unavoidable. In this case, PADMA takes all measures to ensure sustainability.
PADMA takes as little as possible from nature and as much as necessary to supply customers with PADMA original formulas, but also to protect biodiversity. We think about the future, not only of our customers, but also of our planet.
Quality
PADMA relies on trust and safety from the procurement of raw materials to delivery to the customer. Production takes place in accordance with the high quality standards of the pharmaceutical industry at the production facility in Switzerland, which specialises in Tibetan formulations.
PADMA standards are high. Regardless of whether they are medicines or food supplements, PADMA products are manufactured to pharmaceutical quality standards.

Both the selection of raw materials and all production steps are subject to a multi-stage testing and quality assurance process. This includes internal and external laboratory analyses. The herbal raw materials, the active ingredients and the finished product are chemically, physically, biologically and pharmaceutically analysed to ensure that they always meet the strict quality requirements of the Swiss authorities.
PADMA follows the guidelines of good manufacturing practice and respects the needs of customers and a sustainable world. PADMA products are vegan, gluten-free, lactose-free and free from colourings, flavourings and preservatives. Only an absolutely perfect product reaches your hands: pure power of nature in Swiss quality.

Company
Inspiration The story of PADMA begins in 1958 at the ETH Zurich. There, Karl Lutz, a successful Zurich pharmaceutical entrepreneur, attended a lecture on Tibetan medicine. The lecturer, Dr. Cyrill…
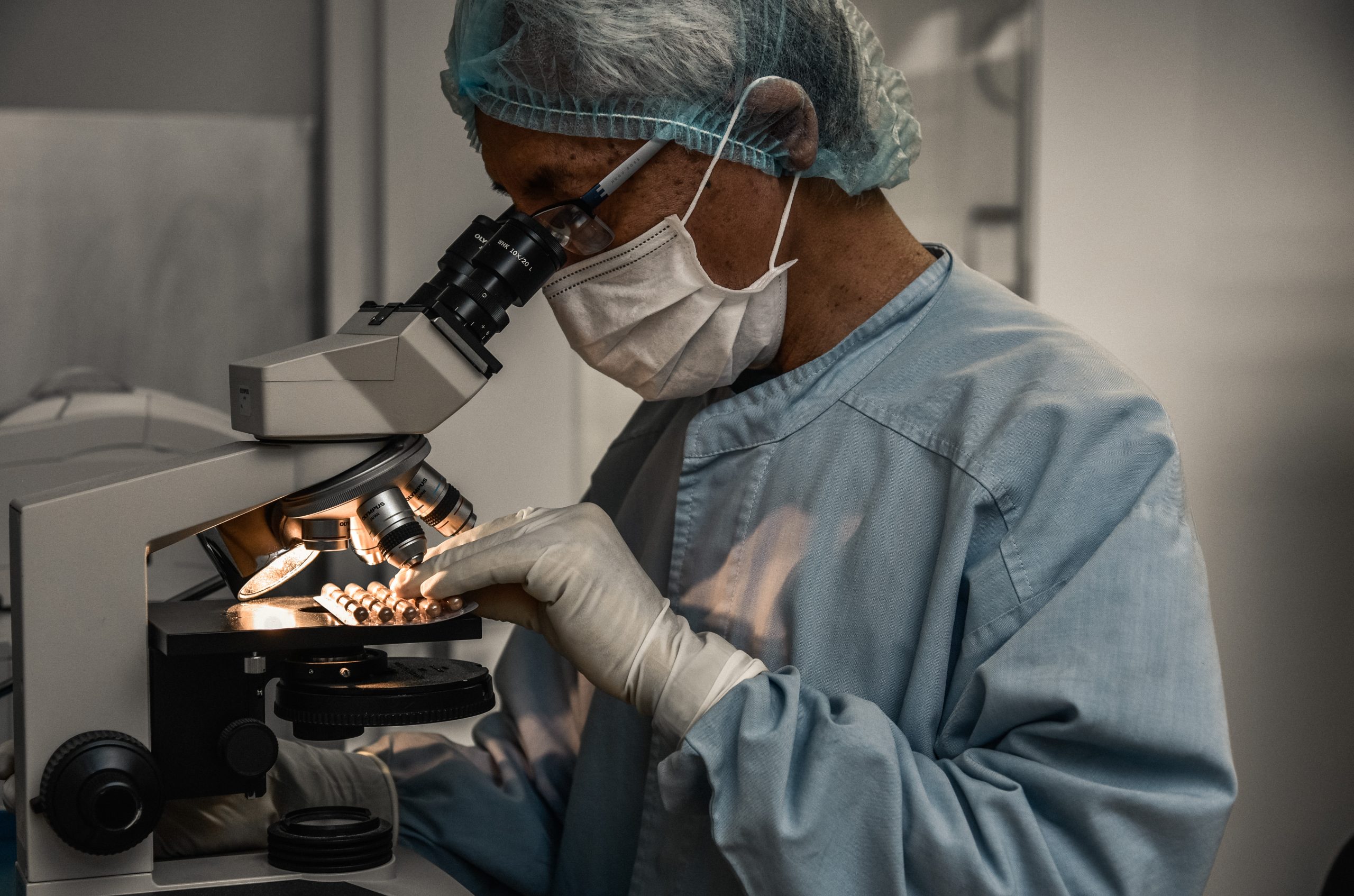
Research
Knowledge builds bridges Research into centuries of old herbal recipes from Tibetan Medicine is the cornerstone of the company’s history. The founder of PADMA, Karl Lutz, was convinced of the meaningfulness…

Social responsibility
People, plants and health are the daily focus of PADMA’s activities. This is based on respectful cooperation at eye level with our customers, employees, suppliers and partners. Shared values inspire…

